Dialysis Filtration
Dialysis Filtration
For dialysis treatment providers, care teams, and patients, the need to reduce treatment time, hospitalizations and medication, while improving blood pressure and hemoglobin profiles, is paramount. Our dialysis filtration solutions for dialysis water offer effective product support for dialysis care, whether in a clinic or at home.
What is dialysis?
Dialysis is a procedure to remove waste products and excess fluid from the blood when the kidneys stop working properly. It often involves diverting blood to a machine to be cleaned.(1)
Who needs dialysis?
When a person’s kidney fails, dialysis is used to keep the body in balance. In chronic or end-stage kidney failure, the kidneys do not get better and the person will need dialysis for the rest of their life. In these cases, dialysis is administered to remove waste, salt, and extra water to prevent them from building up in the body. Treatment also helps the person keep a safe level of certain chemicals in the blood, such as potassium, sodium, and bicarbonate, and helps to control blood pressure.(2)
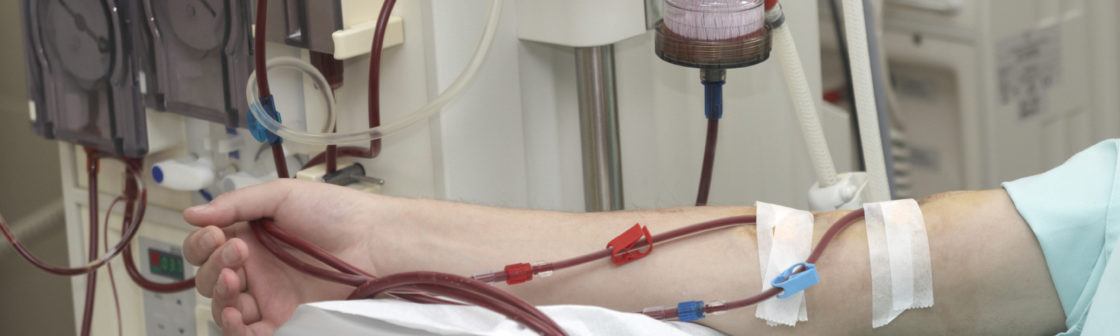
How does a dialysis machine work?
During dialysis treatment, two needles are inserted into a patient’s AV fistula or graft and taped into place. One needle removes blood and transfers it to a dialysis machine called a dialyzer, often referred to as an “artificial kidney”. The dialyzer consists of a series of membranes that act as filters and a special liquid called dialysate. The membranes filter waste products from the blood, which are passed into the dialysate fluid. The used dialysate fluid is pumped out of the dialyzer, and the filtered blood is passed into the patient’s body through the second needle.(1)
What is filtration in dialysis?
Dialysis Water Filtration
Water is a critical input for dialysis where a patient can be exposed to 300 – 600 liters of water per week during hemodialysis treatment. Designed to assist in the production of water that meets or exceeds AAMI standards for dialysis water, our dialysis water filtration seamlessly fits into your central water system or portable DI/RO equipment.
What is dialysis used for?
Dialysis centers provide access to hemodialysis treatment in which smaller waste products, like potassium, creatinine, and excess fluid, in the blood are eliminated by way of a dialyzer as opposed to a kidney. Dialysis water filters retain bacteria, viruses, and endotoxins, preparing water and bicarbonate, for dialysis treatment.
(1) https://www.nhs.uk/conditions/dialysis/
(2) https://www.kidney.org/atoz/content/dialysisinfo#when-dialysis-needed
Want to learn more?
Contact us for product details and additional information
Want to know more?
Reach out for quotes, additional product details, installation questions, and more.
